Impact of HIV-1 Coinfection on Anti-TB Drug Exposure in South African Outpatients
The study investigated the impact of HIV-1 coinfection on the exposure to antibiotics in South African tuberculosis (TB) outpatients.

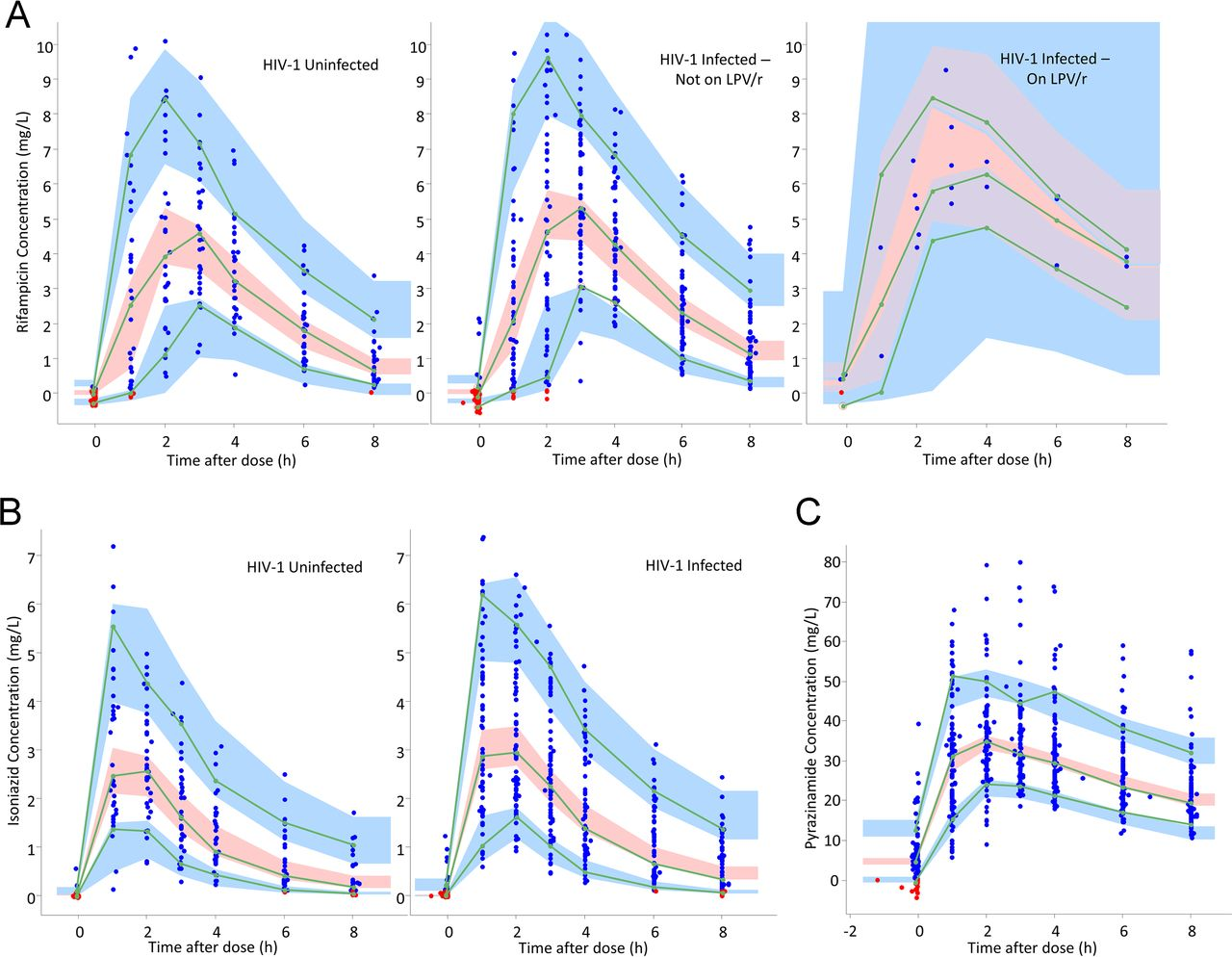
The researchers conducted a prospective pharmacokinetic study involving 119 TB patients, including both HIV-1 coinfected and HIV-uninfected individuals. They measured drug concentrations in plasma and assessed exposure to the drugs using area under the concentration-time curve (AUC) and maximum concentration (Cmax) values.
The results indicated that HIV-1 coinfection did not significantly affect the exposure to rifampin, isoniazid, or pyrazinamide in the study population. Both groups, HIV-1 coinfected and HIV-uninfected, showed similar drug exposures, suggesting that the standard doses of these anti-TB drugs are appropriate for both groups, at least in the outpatient setting studied.
--> These findings suggest that current TB treatment regimens can be effective in HIV-1 coinfected individuals, without the need for dosage adjustments. However, further research is needed to confirm these results in larger and more diverse populations to ensure optimal TB treatment outcomes in HIV-1 coinfected individuals.